The liquefying gases experiment performed by Michael Faraday was one of the most revolutionary achievements in the history of chemistry and physics. Long before modern refrigerators, air conditioners, and cryogenic systems existed, Faraday proved that gases could be transformed into liquids under the right conditions. His experiments changed scientific understanding of matter forever and opened the door to modern refrigeration technology.
During the early 19th century, many scientists believed certain gases could never become liquids. These “permanent gases” seemed impossible to condense because they always remained invisible and difficult to control. Faraday challenged this belief through dangerous and highly creative experiments involving pressure, temperature, and sealed glass tubes.
The liquefying gases experiment not only transformed thermodynamics and gas science but also helped humanity develop cooling technology, industrial refrigeration, cryogenics, and modern refrigeration systems.
This article explores the history, science, equations, experiments, and global impact behind Faraday’s gas liquefaction discoveries.
The Scientific World Before Gas Liquefaction (1800 – 1820)
In the early 1800s, scientists were still learning about the state of matter.
Matter was generally divided into:
- Solids
- Liquids
- Gases
Scientists understood that heating liquids produced gases, but many believed gases could never return to liquid form under normal conditions.
Some gases like steam could condense easily, but others such as chlorine gas and ammonia seemed impossible to liquefy.
At this time, gas laws were becoming important in chemistry and physics.
One famous equation was Boyle’s Law:
Where:
- = Pressure
- = Volume
- = Constant
This law showed that increasing pressure reduces gas volume.
Scientists suspected high pressure might eventually force gases into liquid form.
Michael Faraday and Experimental Chemistry
Michael Faraday became fascinated with gases and chemical reactions during his work at the Royal Institution.
Faraday was already famous for experimental skill and scientific curiosity. His chemistry research included:
- Electrochemistry
- Gas analysis
- Chemical reactions
- Pressure experiments
Faraday believed gases were not fundamentally different from liquids.
He suspected that under the right combination of temperature and pressure, gases could condense into liquids.
This idea led directly to the liquefying gases experiment.
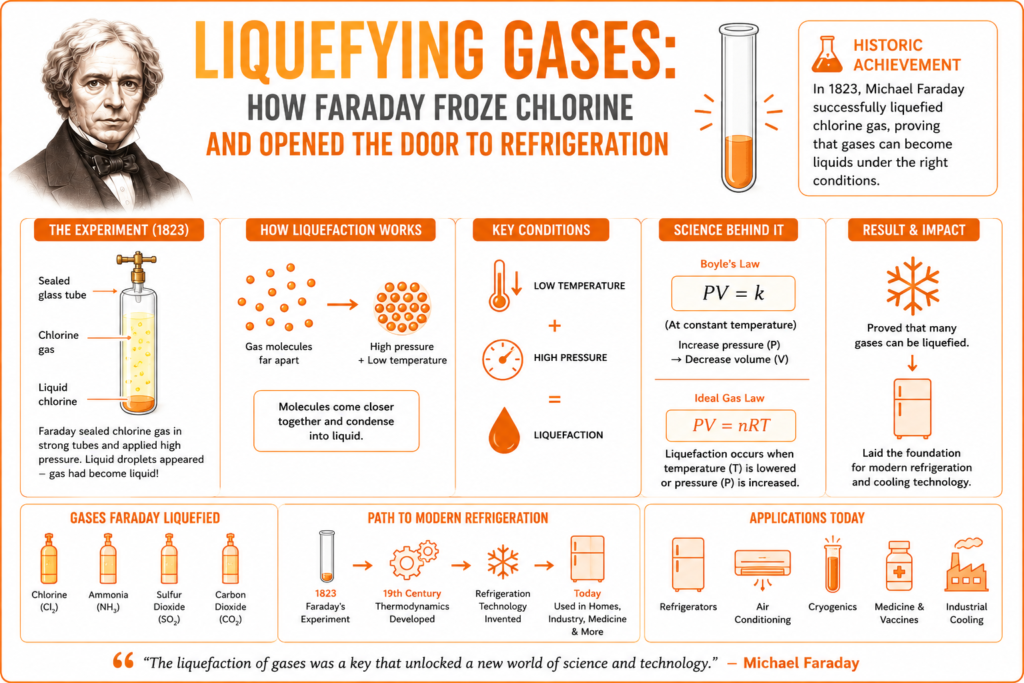
The Famous Chlorine Experiment (1823)
In 1823, Faraday performed one of the most important chemistry experiments in history.
He sealed chlorine gas inside strong glass tubes along with chemicals that produced pressure naturally.
Under high pressure and lower temperature conditions, something remarkable happened.
Liquid droplets suddenly appeared inside the tube.
Faraday had successfully liquefied chlorine gas.
This experiment shocked the scientific world.
For the first time, scientists clearly observed chlorine existing as a liquid.
The liquefying gases experiment proved gases and liquids were connected through physical conditions rather than being completely separate substances.
Understanding Gas Liquefaction
Gas liquefaction occurs when gas molecules lose enough thermal energy to condense into liquid form.
This process usually requires:
- Lower temperature
- Higher pressure
The relationship between pressure, volume, and temperature is described by the Ideal Gas Law:
Where:
- = Pressure
- = Volume
- = Number of moles
- = Gas constant
- = Temperature
When temperature decreases or pressure increases sufficiently, gas molecules move closer together and condense.
This phase transition became central to modern thermodynamics.
Phase Transition and States of Matter
Faraday’s experiments revealed important truths about phase transition.
Matter can change between:
- Solid
- Liquid
- Gas
These transitions depend on thermal energy and pressure conditions.
For example:
This process is called condensation.
The reverse process:
is evaporation.
Faraday’s experiments showed that gases were simply another form of matter rather than mysterious permanent substances.
Vapor Pressure and Condensation
Vapor pressure became one of the key ideas in gas liquefaction science.
Vapor pressure is the pressure exerted by gas molecules escaping from a liquid surface.
Condensation occurs when gas pressure exceeds vapor pressure at a given temperature.
Faraday carefully controlled these conditions inside sealed tubes.
His experiments demonstrated how pressure and cooling work together during liquefaction.
These ideas later became essential in refrigeration engineering.
Critical Temperature Explained
One of the most important concepts related to liquefying gases experiment is critical temperature.
Critical temperature is the highest temperature at which a gas can become liquid regardless of pressure.
Above this temperature:
- Liquefaction becomes impossible
Below it:
- Pressure can condense gas into liquid form
Faraday’s work helped scientists later understand critical points and phase diagrams more deeply.
This knowledge became essential for cryogenics and cooling technology.
Heat of Vaporization and Thermal Energy
Liquefaction depends heavily on heat transfer.
When gases condense into liquids, they release heat called latent heat or heat of vaporization.
The heat equation is:
Where:
- = Heat energy
- = Mass
- = Latent heat of vaporization
Removing thermal energy slows gas molecules and allows condensation.
Modern refrigeration systems still rely on these same principles discovered through Faraday’s experiments.
Sealed Tubes and High Pressure Experiments
Faraday’s sealed tubes were extremely dangerous.
Many exploded because of intense pressure buildup.
Yet Faraday continued experimenting carefully with gases such as:
- Chlorine
- Ammonia
- Sulfur dioxide
- Carbon dioxide
These high-pressure experiments required extraordinary scientific courage and precision.
Faraday’s laboratory skill helped him succeed where many others failed.
Ammonia Liquefaction and Refrigeration
Faraday also worked with ammonia liquefaction.
Ammonia later became one of the most important refrigeration gases in history.
Modern refrigerators operate using vapor compression cycles involving:
- Compression
- Condensation
- Expansion
- Evaporation
These systems depend directly on gas liquefaction principles first demonstrated by Faraday.
Without the liquefying gases experiment, refrigeration technology would have developed far more slowly.
The Birth of Refrigeration Technology (1850 – 1900)
Faraday’s discoveries eventually transformed refrigeration engineering.
Scientists realized gases could absorb and release heat efficiently during phase transitions.
This led to the invention of:
- Refrigerators
- Ice making machines
- Air conditioning systems
- Industrial cooling systems
Refrigeration became essential for:
- Food preservation
- Medicine storage
- Industrial manufacturing
- Transportation
Faraday’s experiments helped change everyday life permanently.
Thermodynamics and Faraday’s Influence
Faraday’s work strongly influenced thermodynamics.
Thermodynamics studies heat, energy, and physical systems.
The First Law of Thermodynamics states:
Where:
- = Internal energy change
- = Heat added
- = Work done
Gas liquefaction became an important example of energy transformation.
Faraday’s experiments helped scientists understand how heat and pressure affect matter.
Cryogenics and Modern Science
Modern cryogenics developed partly from Faraday’s gas experiments.
Cryogenics studies matter at extremely low temperatures.
Today, liquefied gases are used in:
- MRI machines
- Rocket fuels
- Superconductors
- Medical technology
- Scientific research
Liquid nitrogen and liquid helium are now common scientific tools.
Faraday’s early experiments opened the door to these advanced technologies.
Faraday and Industrial Chemistry
Faraday’s chemistry work extended beyond gases.
His research also included faraday discovery of benzene, electrochemistry, and materials science.
These combined discoveries transformed industrial chemistry during the Industrial Revolution.
Faraday’s work connected chemistry, thermodynamics, and physics into one powerful scientific framework.
Electromagnetism and Scientific Legacy
Although Faraday became famous for electricity later, his chemistry research was equally important.
His later discoveries in electromagnetic induction and electricity transformed civilization in different ways.
Together, Faraday’s work shaped modern science more deeply than almost any other scientist of his era.
Boyle’s Law and Gas Compression
Faraday’s liquefaction experiments strongly relied on Boyle’s Law.
The equation:
shows how increasing pressure reduces gas volume.
As gas molecules move closer together:
- Intermolecular attraction increases
- Condensation becomes possible
This principle remains central in refrigeration systems today.
Phase Diagrams and Modern Chemistry
Modern chemistry uses phase diagrams to study matter behavior.
Phase diagrams show relationships between:
- Pressure
- Temperature
- State of matter
Faraday’s experiments helped scientists understand these relationships experimentally.
Phase diagrams now guide industries involving refrigeration, metallurgy, and cryogenics.
Why Faraday’s Gas Experiments Still Matter
The liquefying gases experiment remains historically important because it transformed scientific understanding completely.
Without Faraday’s work:
- Refrigerators might not exist
- Air conditioning would develop later
- Cryogenic science would slow dramatically
- Food preservation systems would suffer
Modern civilization depends heavily on controlled cooling technology.
Faraday helped make that possible.
Modern Applications of Gas Liquefaction (1900 – Present)
Today, gas liquefaction technologies operate worldwide.
Applications include:
- LNG fuel storage
- Refrigeration systems
- Air conditioning
- Medical cooling systems
- Industrial gas transport
- Space technology
Modern cooling systems still rely on the same condensation principles Faraday demonstrated nearly two centuries ago.
Faraday’s Scientific Legacy
Faraday’s scientific legacy spans chemistry, physics, and engineering.
His discoveries influenced:
- Refrigeration
- Thermodynamics
- Electrochemistry
- Cryogenics
- Modern industry
Faraday showed that scientific curiosity and experimentation could transform civilization permanently.
FAQs About Liquefying Gases Experiment
What was Faraday’s liquefying gases experiment?
Faraday’s experiment showed that gases like chlorine could become liquids under high pressure and low temperature.
Why was liquefying chlorine important?
It proved gases and liquids are related states of matter rather than completely separate substances.
What is gas liquefaction?
Gas liquefaction is the process of converting gas into liquid using pressure and cooling.
How did Faraday help refrigeration technology?
His experiments established the scientific principles behind refrigeration and cooling systems.
What is critical temperature?
Critical temperature is the highest temperature at which a gas can become liquid.
What industries use gas liquefaction today?
Gas liquefaction is used in refrigeration, cryogenics, medicine, fuel transport, and industrial cooling.
Conclusion
The liquefying gases experiment performed by Michael Faraday was one of the greatest breakthroughs in scientific history. By successfully liquefying chlorine gas, Faraday proved that gases could transform into liquids under the right conditions of pressure and temperature.
This discovery completely changed humanity’s understanding of matter, thermodynamics, and energy transfer. More importantly, it opened the path toward refrigeration, cryogenics, air conditioning, and modern cooling technologies that millions depend on every day.
Faraday’s experiments connected chemistry, thermodynamics, and industrial engineering into one powerful scientific framework. His discoveries continue influencing science, medicine, transportation, and energy systems worldwide.
The impact of Faraday’s liquefaction research on civilization is as revolutionary as how ancient greek scientists changed modern science, proving once again that bold scientific curiosity can reshape the future of humanity forever.