The faraday discovery of benzene was one of the most important breakthroughs in the history of chemistry. Although Michael Faraday is often remembered for electricity and magnetism, he was also a brilliant chemist whose work transformed modern industrial science. His discovery of benzene helped lay the foundation for organic chemistry, petrochemicals, plastics, medicines, dyes, fuels, and countless industrial products used today.
In 1825, Faraday isolated a strange hydrocarbon from compressed illumination gas. At the time, nobody realized how important this new substance would become. Decades later, benzene became one of the central molecules in chemical synthesis and modern industrial chemistry.
Today, faraday discovery of benzene continues influencing science, manufacturing, pharmaceuticals, energy systems, and materials engineering worldwide.
This article explores the history, experiments, chemistry, equations, scientific importance, and modern applications connected to Faraday’s discovery of benzene.
Michael Faraday as a Chemist (1810 – 1830)
Most people associate Michael Faraday with electricity and magnetism. However, during the early years of his scientific career, Faraday worked extensively in chemistry.
At the Royal Institution, he performed experiments involving:
- Gases
- Metals
- Acids
- Chemical reactions
- Hydrocarbon molecules
Faraday developed exceptional laboratory skills through experimentation and observation.
His success largely came from michael faraday self education, which allowed him to study chemistry independently despite lacking formal university training.
Faraday believed chemistry could reveal the hidden structure of matter itself.
This belief eventually led him to discover benzene.
The Scientific World Before Benzene
In the early 19th century, organic chemistry was still developing.
Scientists knew very little about:
- Carbon compounds
- Molecular structure
- Hydrocarbon molecules
- Chemical bonding
Coal gas had recently become important for street lighting and industrial use.
This illumination gas contained many unknown chemicals produced during coal processing.
Chemists began analyzing coal gas carefully to identify useful compounds.
Faraday became fascinated by these mysterious substances.
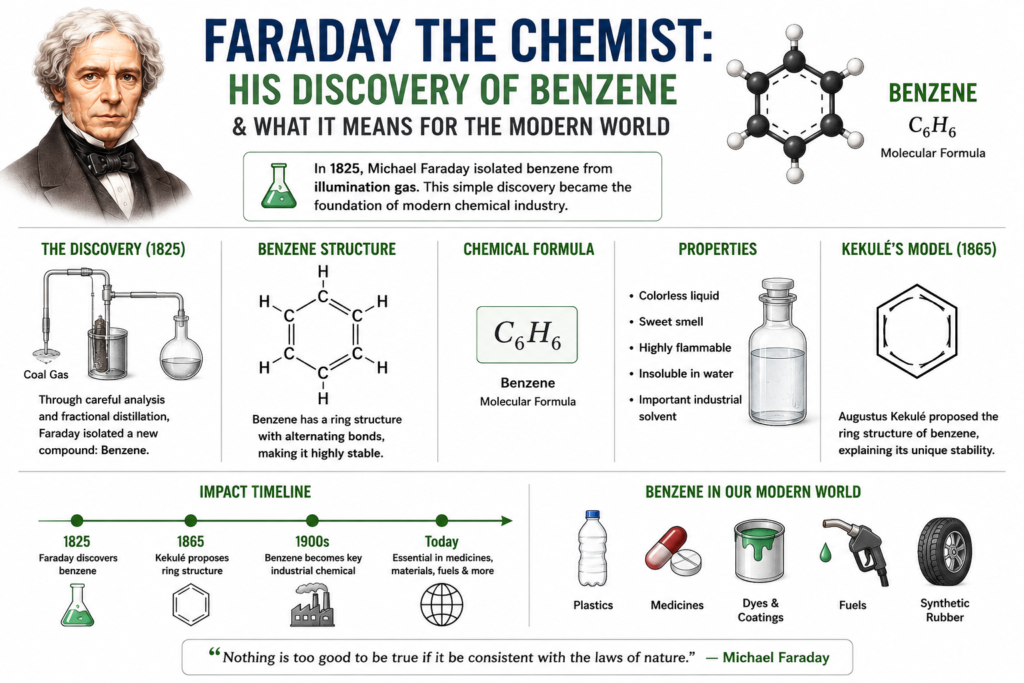
The Discovery of Benzene (1825)
In 1825, Faraday analyzed oily residues found in compressed illumination gas cylinders.
Using careful chemical analysis and fractional distillation techniques, he isolated a colorless liquid with unusual properties.
Faraday initially called the compound:
“Bicarburet of hydrogen”
This substance later became known as benzene.
The molecular formula of benzene is:
This simple formula puzzled scientists for decades because it did not behave like normal hydrocarbons.
The faraday discovery of benzene eventually became one of the most important moments in organic chemistry.
Understanding the Formula of Benzene
The formula:
means benzene contains:
- 6 carbon atoms
- 6 hydrogen atoms
Scientists noticed something unusual.
Most hydrocarbons with so few hydrogen atoms should behave very reactively. However, benzene showed remarkable molecular stability.
This mystery fascinated chemists for many years.
Later, scientists discovered benzene forms a carbon ring structure with special bonding behavior.
This discovery transformed chemical structure theory forever.
Faraday’s Experimental Method
Faraday relied heavily on experimentation rather than theoretical speculation.
He used techniques including:
- Fractional distillation
- Solvent extraction
- Combustion analysis
- Vapor studies
These methods helped him isolate pure compounds from complex gas mixtures.
Faraday’s careful observations allowed him to identify benzene as a completely new substance.
His work demonstrated extraordinary scientific precision for the time.
Coal Gas and Industrial Chemistry
Coal gas played a major role in Faraday’s research.
Coal gas was produced by heating coal without oxygen.
The process created:
- Hydrogen
- Methane
- Carbon monoxide
- Hydrocarbon vapors
Faraday studied these substances extensively.
The discovery of benzene showed that industrial byproducts could contain valuable chemical compounds.
This realization transformed industrial chemistry dramatically.
Chemical Reactions and Benzene
Benzene behaves differently from many ordinary hydrocarbons.
Instead of reacting through simple addition reactions, benzene prefers substitution reactions.
For example:
In this reaction:
- One hydrogen atom is replaced by bromine
- The benzene ring remains stable
This unusual behavior later became central to aromatic hydrocarbons chemistry.
Augustus Kekulé and Benzene Structure (1865)
For decades, scientists struggled to explain benzene’s structure.
In 1865, chemist Augustus Kekulé proposed the famous carbon ring model.
According to Kekulé:
- Benzene forms a hexagonal ring
- Carbon atoms connect through alternating double bonds
The simplified structure can be represented as:
Modern chemistry later showed that benzene actually contains delocalized electrons shared across the ring.
This electron sharing explains benzene’s molecular stability.
The faraday discovery of benzene eventually became one of the foundations of structural chemistry.
Covalent Bonds and Aromatic Stability
Benzene’s unique behavior comes from covalent bonds and electron delocalization.
Each carbon atom shares electrons with neighboring atoms.
Modern quantum chemistry describes benzene using resonance structures.
The average bond length in benzene is:
This bond length lies between single and double carbon bonds.
Benzene’s stability arises because electrons spread evenly around the ring.
This special arrangement became central to aromatic hydrocarbons chemistry.
Benzene and Organic Chemistry
The faraday discovery of benzene transformed organic chemistry permanently.
Chemists realized carbon compounds could form highly organized molecular structures.
Benzene later became essential for producing:
- Plastics
- Medicines
- Synthetic fibers
- Dyes
- Explosives
- Detergents
- Rubber products
Modern chemical synthesis depends heavily on benzene derivatives.
Faraday’s discovery opened the door to entire industrial sectors.
Benzene and Petrochemicals (1900 – Present)
Today, benzene remains one of the most important petrochemicals worldwide.
Petroleum refining produces benzene through catalytic reforming processes.
Modern industries use benzene to manufacture:
- Styrene
- Phenol
- Nylon
- Polyurethane
- Pharmaceuticals
The chemical industry relies heavily on aromatic compounds first connected to Faraday’s work.
Global industrial chemistry would look completely different without benzene.
Benzene and Chemical Equations
One common industrial reaction involving benzene is nitration:
This reaction produces nitrobenzene, which later becomes useful for dyes and chemicals.
Another important reaction is hydrogenation:
This converts benzene into cyclohexane.
Such reactions became essential in industrial manufacturing.
Faraday’s Broader Chemical Research
Faraday’s scientific work extended beyond benzene.
He also studied:
- Electrochemistry
- Gas liquefaction
- Magnetism
- Optics
His work later contributed to faraday’s law of electrolysis, which connected electricity and chemical reactions mathematically.
Faraday’s contributions helped unify chemistry and physics into interconnected sciences.
Benzene and Modern Medicine
Modern medicine relies heavily on benzene derivatives.
Many drugs contain aromatic ring structures derived from benzene chemistry.
Examples include:
- Aspirin
- Paracetamol
- Antibiotics
- Antihistamines
Benzene based compounds became essential for pharmaceutical science.
Faraday’s discovery indirectly influenced modern healthcare globally.
Benzene and Materials Science
Modern materials science also depends on benzene chemistry.
Benzene derivatives help produce:
- Plastics
- Epoxy resins
- Synthetic rubber
- Carbon fibers
- Paints and coatings
These materials power industries including:
- Construction
- Transportation
- Aerospace
- Electronics
Faraday’s discovery became one of the foundations of modern manufacturing.
Environmental and Health Concerns
Although benzene is industrially important, scientists later discovered health risks associated with long term exposure.
Benzene can affect:
- Bone marrow
- Blood production
- Human health
Modern industries carefully regulate benzene handling and emissions.
This demonstrates how scientific discoveries often bring both benefits and responsibilities.
Faraday and Scientific Thinking
Faraday’s work demonstrated the power of careful experimentation.
He discovered benzene without advanced theoretical chemistry.
Instead, he relied on:
- Observation
- Precision
- Curiosity
- Laboratory skill
His scientific imagination later influenced ideas related to field theory and lines of force and many other discoveries.
Faraday proved that experimentation could reveal entirely new worlds of knowledge.
Relation Between Benzene and Modern Physics
Faraday’s discoveries in chemistry and physics were deeply connected.
His studies in electricity later contributed to electromagnetic induction and electrical science.
These combined discoveries transformed modern technology and industrial civilization.
Few scientists have influenced both chemistry and physics as deeply as Faraday.
Industrial Revolution and Benzene
The Industrial Revolution accelerated rapidly because of chemical discoveries like benzene.
Industries gained access to:
- Synthetic dyes
- Chemical fuels
- Advanced materials
- Pharmaceutical compounds
Benzene became one of the building blocks of modern industrial chemistry.
Faraday’s discovery influenced global economic development for generations.
Why the Discovery of Benzene Still Matters
Today, faraday discovery of benzene remains central to chemistry education and industrial science.
Without benzene chemistry:
- Modern plastics would disappear
- Pharmaceutical industries would suffer
- Petrochemical systems would collapse
- Synthetic materials would become limited
Faraday’s discovery still shapes daily life worldwide.
Faraday’s Scientific Legacy
Faraday’s scientific legacy extends far beyond electricity.
His chemistry research transformed scientific understanding of matter itself.
He inspired generations of chemists, physicists, and engineers through his discoveries and humility.
Faraday showed that science progresses through curiosity, discipline, and experimentation.
FAQs About Faraday Discovery of Benzene
What did Michael Faraday discover in chemistry?
Faraday discovered benzene and made major contributions to electrochemistry and gas research.
When was benzene discovered?
Michael Faraday discovered benzene in 1825.
What is the formula of benzene?
The chemical formula of benzene is:
Why is benzene important?
Benzene is essential in plastics, medicines, fuels, dyes, and industrial chemistry.
What are aromatic hydrocarbons?
Aromatic hydrocarbons are stable ring shaped carbon compounds like benzene.
Who explained the structure of benzene?
Augustus Kekulé proposed the ring structure of benzene in 1865.
Conclusion
The faraday discovery of benzene was one of the most important breakthroughs in the history of chemistry. Michael Faraday transformed industrial science by isolating a mysterious hydrocarbon that later became central to organic chemistry and petrochemical industries.
From plastics and medicines to fuels and advanced materials, benzene chemistry powers countless modern technologies. Faraday’s careful experiments revealed the extraordinary complexity and beauty of carbon compounds long before modern molecular theory fully developed.
His discoveries helped shape chemistry, physics, medicine, and industrial manufacturing on a global scale. The impact of Faraday’s work remains visible in nearly every aspect of modern life.
The revolutionary influence of benzene chemistry on civilization is as powerful as how ancient greek scientists changed modern science, proving once again that curiosity driven experiments can transform the future of humanity forever.