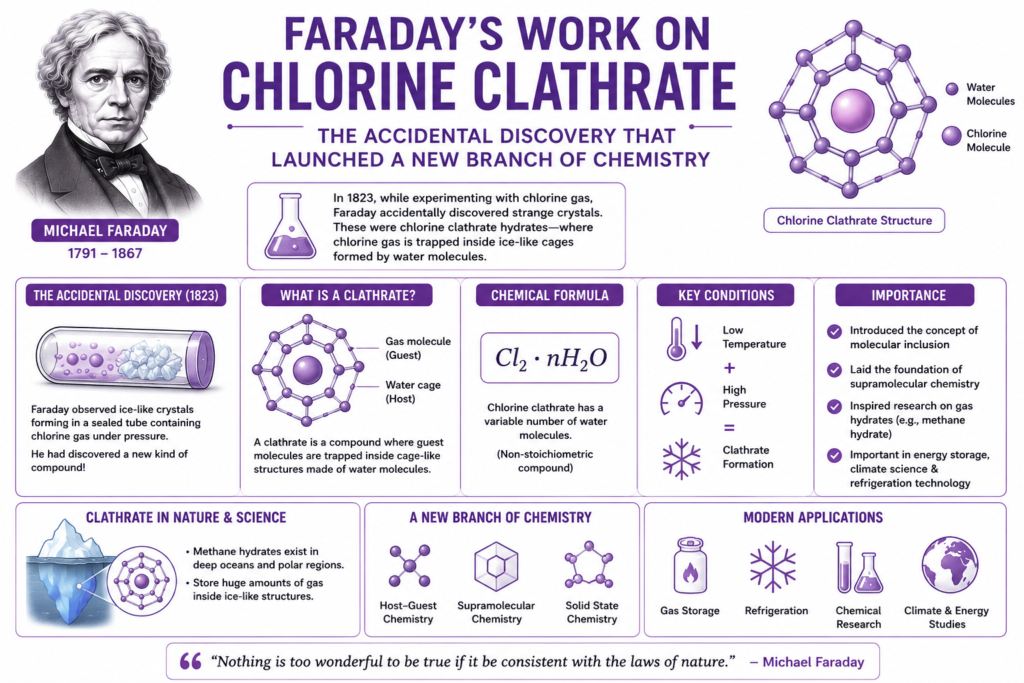
The chlorine clathrate discovery made by Michael Faraday was one of the most unusual and important accidental discoveries in chemistry. While experimenting with chlorine gas during the early 19th century, Faraday observed strange crystal formations that did not behave like ordinary compounds. At first, scientists did not fully understand what these materials were. Later, these mysterious crystals became known as clathrate hydrates and eventually launched entirely new areas of chemistry and material science.
Today, chlorine clathrate research influences supramolecular chemistry, gas storage technology, climate science, cryogenics, and energy research. Faraday’s accidental discovery revealed that molecules could trap other molecules inside cage-like structures without traditional chemical bonding.
The chlorine clathrate discovery completely changed scientific understanding of molecular inclusion and solid state chemistry.
This article explores the history, experiments, chemistry, equations, scientific importance, and modern applications connected to Faraday’s discovery of chlorine clathrates.
The Scientific World Before Clathrate Chemistry (1800 – 1823)
In the early 19th century, chemistry was still developing rapidly.
Scientists understood basic compounds and chemical reactions, but they knew very little about:
- Molecular structures
- Crystal formation
- Gas hydrates
- Host-guest chemistry
- Non-stoichiometric compounds
Most chemists believed compounds formed only through direct chemical bonding between atoms.
The idea that molecules could physically trap other molecules inside crystal structures seemed impossible.
At this time, Michael Faraday was conducting dangerous experiments involving chlorine gas and pressure systems.
During these experiments, he accidentally discovered something extraordinary.
Michael Faraday and Experimental Chemistry
Faraday was famous for careful experimentation and scientific curiosity.
His chemistry work included studies involving:
- Gases
- Electrochemistry
- Crystal formation
- Condensation reactions
- Pressure experiments
Faraday’s laboratory methods were extremely precise.
Much of his success came through michael faraday self education, which allowed him to master chemistry despite limited formal schooling.
Faraday believed observation was the foundation of scientific truth.
This mindset helped him notice unusual crystal formations others might have ignored.
The Chlorine Experiments (1823)
In 1823, Faraday performed experiments involving chlorine gas inside sealed glass tubes.
These experiments were originally part of his famous liquefying gases experiment research.
Faraday compressed chlorine gas under controlled pressure and temperature conditions.
During one experiment, he observed strange ice-like crystals forming inside the tube.
These crystals were unusual because they appeared solid yet contained trapped chlorine gas molecules.
At first, Faraday believed the crystals were simply frozen chlorine compounds.
Later, scientists realized these materials were chlorine hydrate clathrates.
The chlorine clathrate discovery became one of the earliest examples of molecular inclusion chemistry.
What Is a Clathrate?
A clathrate is a compound where one type of molecule traps another molecule inside a cage-like structure.
The word “clathrate” comes from the Latin word clathratus, meaning enclosed or protected by bars.
In chlorine hydrate:
- Water molecules form crystal cages
- Chlorine molecules become trapped inside
Importantly, chlorine molecules are not chemically bonded strongly to water molecules.
Instead, they are physically enclosed.
This unusual arrangement became the foundation of supramolecular chemistry.
Understanding Chlorine Hydrate
Chlorine hydrate is one of the earliest known gas hydrates.
Its approximate composition is often represented as:
Where:
- = Chlorine gas molecule
- = Water molecules
- = Variable number of water molecules
The value of n is not always fixed because clathrates are non-stoichiometric compounds.
This unusual behavior puzzled early chemists.
Unlike ordinary compounds, clathrates do not follow simple atomic ratios.
Cage Like Structures and Molecular Inclusion
The chlorine clathrate discovery revealed a completely new type of molecular structure.
Water molecules arrange themselves into lattice structures forming cages.
Inside these cages, gas molecules become trapped.
This process is called molecular inclusion.
The crystal structure stabilizes through intermolecular forces rather than normal covalent bonds.
Modern chemistry describes these interactions using host-guest chemistry concepts.
- Host molecules form the cage
- Guest molecules occupy the interior space
This discovery transformed understanding of chemical anomalies and solid state chemistry.
Crystal Structure and Water Molecules
The crystal lattice inside clathrate hydrates resembles ice structures.
Water molecules connect through hydrogen bonding.
The hydrogen bond interaction can be represented simply as:
These bonds create stable cage frameworks.
The cages then trap chlorine molecules inside open spaces.
This arrangement gives clathrates unique physical properties.
The chlorine clathrate discovery helped scientists understand how water molecules organize themselves into complex structures.
Pressure Temperature Conditions in Clathrate Formation
Clathrate formation depends strongly on pressure-temperature conditions.
Generally:
- Lower temperatures favor clathrate formation
- Higher pressures stabilize trapped gases
Faraday’s experiments unintentionally created the perfect conditions for chlorine hydrate formation.
The relationship between pressure and gas behavior follows the Ideal Gas Law:
Where:
- = Pressure
- = Volume
- = Number of moles
- = Gas constant
- = Temperature
By controlling pressure and cooling, Faraday accidentally produced clathrate crystals.
Chemical Bonding and Clathrates
Traditional chemistry focused mainly on covalent bonds and ionic compounds.
Clathrates introduced a different idea.
The trapped gas molecules are held through weak intermolecular forces rather than direct chemical bonding.
These include:
- Van der Waals forces
- Dipole interactions
- Hydrogen bonding stabilization
This discovery expanded chemistry beyond ordinary molecular structures.
Modern supramolecular chemistry later developed from these ideas.
Clathrates and Thermodynamics
Clathrate formation also involves thermodynamics.
The stability of clathrates depends on energy balance.
The Gibbs free energy equation is:
Where:
- = Free energy change
- = Enthalpy change
- = Temperature
- = Entropy change
Clathrates form when the overall energy state becomes favorable under certain conditions.
This thermodynamic behavior became important in modern chemistry and materials science.
Methane Hydrate and Modern Energy Research
Faraday’s accidental discovery later inspired research into methane hydrate.
Methane hydrate is another clathrate where methane molecules become trapped inside water cages.
The approximate formula is:
Massive methane hydrate deposits exist beneath oceans and Arctic regions.
Scientists now study these deposits as possible future energy sources.
The chlorine clathrate discovery eventually influenced global energy research dramatically.
Supramolecular Chemistry and Host Guest Systems
Modern supramolecular chemistry grew partly from clathrate research.
This branch of chemistry studies systems held together through weak intermolecular interactions.
Examples include:
- Molecular cages
- DNA interactions
- Protein folding
- Drug delivery systems
Host-guest chemistry became essential in nanotechnology and pharmaceutical science.
Faraday’s accidental discovery helped launch this entire scientific field.
Clathrates and Refrigeration Science
Clathrates also influenced refrigeration research.
Gas hydrates absorb and release heat during formation and decomposition.
This behavior became useful in cooling systems and thermal energy storage.
The chlorine clathrate discovery helped scientists better understand phase transitions and crystal stability.
Faraday’s work indirectly influenced refrigeration engineering and cryogenic science.
Solid State Chemistry and Crystal Science (1900 – Present)
Modern solid state chemistry studies materials with organized crystal structures.
Clathrates became important because they behave differently from ordinary solids.
Scientists discovered clathrates can:
- Trap gases efficiently
- Store energy
- Stabilize reactive molecules
Modern materials science continues using clathrate principles for advanced technologies.
Faraday’s discovery remained scientifically important long after his lifetime.
Clathrates and Climate Science
Methane hydrate research also became important in climate science.
Methane is a powerful greenhouse gas.
Scientists worry that warming oceans could release methane trapped inside hydrate structures.
This could influence global climate systems significantly.
Faraday’s accidental chemistry experiment eventually became connected to environmental science centuries later.
Faraday’s Broader Scientific Work
Faraday’s chemistry research extended beyond clathrates.
He also worked on:
- Electrochemistry
- Gas liquefaction
- Magnetism
- Organic chemistry
His discoveries later contributed to faraday’s law of electrolysis and many other scientific breakthroughs.
Faraday showed extraordinary versatility across scientific disciplines.
Relation Between Chemistry and Physics
Faraday’s work connected chemistry and physics deeply.
His later discoveries involving electromagnetic induction transformed electrical science while his chemistry experiments transformed molecular science.
Few scientists influenced both fields as powerfully as Faraday.
His work demonstrated the unity of natural science.
Molecular Stability and Chemical Anomalies
Clathrates fascinated scientists because they behaved unlike ordinary compounds.
Their unusual properties included:
- Variable composition
- Weak molecular interactions
- Ice-like crystals
- Gas trapping ability
These chemical anomalies challenged traditional chemistry theories.
Faraday’s observations helped scientists realize matter could organize itself in surprisingly complex ways.
Why Chlorine Clathrate Discovery Still Matters
The chlorine clathrate discovery remains important today because it opened entirely new areas of science.
Without clathrate research:
- Gas storage technologies would advance more slowly
- Supramolecular chemistry would develop later
- Cryogenic science would suffer
- Climate hydrate studies would remain limited
Faraday’s accidental discovery still influences modern research worldwide.
Faraday’s Scientific Legacy
Faraday’s scientific legacy extends across chemistry, physics, engineering, and materials science.
His discoveries influenced:
- Molecular chemistry
- Electrochemistry
- Refrigeration science
- Gas storage technology
- Modern materials research
Faraday proved that careful observation and curiosity could reveal entirely new branches of science.
FAQs About Chlorine Clathrate Discovery
What is chlorine clathrate?
Chlorine clathrate is a hydrate where chlorine gas molecules become trapped inside water crystal cages.
Who discovered chlorine clathrate?
Michael Faraday discovered chlorine hydrate accidentally during experiments in 1823.
What are clathrate hydrates?
Clathrate hydrates are crystal structures where water molecules trap gas molecules inside cage-like structures.
Why is the chlorine clathrate discovery important?
It launched new scientific fields including supramolecular chemistry and gas hydrate research.
What is host-guest chemistry?
Host-guest chemistry studies systems where one molecule traps another inside a molecular structure.
What modern applications use clathrate science?
Clathrate research is used in energy storage, refrigeration, climate science, and materials engineering.
Conclusion
The chlorine clathrate discovery made by Michael Faraday was one of the most remarkable accidental breakthroughs in scientific history. While studying chlorine gas and pressure systems, Faraday unknowingly revealed an entirely new type of molecular structure based on trapped gas molecules inside crystal cages.
This discovery transformed chemistry by introducing ideas that later became central to supramolecular chemistry, host-guest systems, gas storage science, and materials engineering. Modern research into methane hydrates, energy systems, refrigeration, and climate science continues building upon principles connected to Faraday’s early experiments.
Faraday’s accidental observations demonstrated the extraordinary power of scientific curiosity and careful experimentation. His work showed that even unexpected laboratory results can reshape human understanding of nature forever.
The scientific importance of chlorine clathrate discovery is as revolutionary as how ancient greek scientists changed modern science, proving once again that accidental discoveries can transform the future of civilization forever.